Ultraviolet germicidal irradiance (UVGI) has the ability to make buildings and indoor spaces healthier for their occupants by significantly reducing the spread of COVID-19 and future airborne respiratory viruses.
Learning Objectives
- Describe the mechanism of spread of airborne respiratory pathogens.
- Explain how UVGI can mitigate the spread of airborne infectious disease.
- Describe the process of simulating UVGI disinfection via UV radiometry and CFD and how UVGI technologies are best applied.
With the world still in the throes of the COVID-19 pandemic, it may be tempting to see the spread of the disease caused by the SARS-CoV-2 pathogen as a singular event. The truth is major viral disease spread is a common motif in recent history. Doctors David Morens and Anthony Fauci of the National Institute of Allergy and Infectious Diseases describe us entering a pandemic era, in which we are increasingly at risk from outbreaks of novel pathogens for which we have little or no acquired immunity.
A recent survey of epidemics since 1600 estimates the likelihood of a novel pathogen sweeping through the world’s population at roughly 2% annually. Given the speed at which novel airborne pathogens such as COVID-19 can spread through an unimmunized population, we may not recognize future pandemics until they are already upon us. It is critical engineers to include infection risk mitigation measures in the design of the built environment.
Identifying how to best respond to the future emergence of novel and highly infectious pathogens requires a better understanding of the mechanisms of disease and the vectors for infection. The chief concern for epidemiologists and public health officials is the spread of viral disease via upper respiratory pathways. Be it a novel strain of influenza or a more virulent variant of the coronavirus, airborne spread through respiratory pathways is the primary route future pandemics will take. Mitigating the spread will be critical in the development of effective strategies to limit infections.

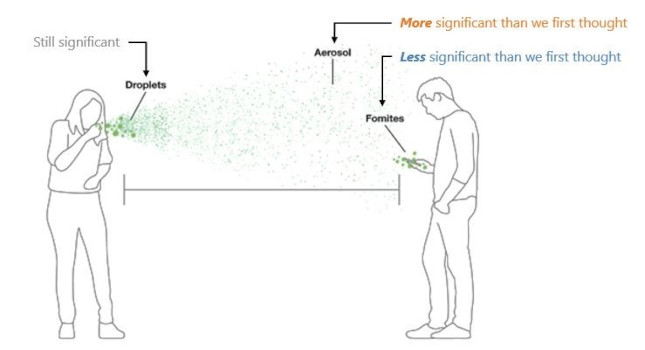
For much of 2020, both the Centers for Disease Control and Prevention and the World Health Organization maintained COVID-19 infections came predominantly from larger infectious respiratory droplets that fell quickly and were deposited within 6 feet of the infected individual without remaining in the air for long. However, the collected evidence has emerged to suggest that they were mistaken.
On July 6, 2020, 239 scientists from 32 countries penned an open letter urging the WHO and other international public health organizations to address the potential the COVID-19 pathogen is airborne. Specifically, infectious particles remain aloft in room air volumes for periods ranging from three hours (according to some studies) up to 12 to 16 hours.
The growing body of published research suggests the COVID-19 pathogen and its subsequent mutational variants are more easily transmitted and retain infectivity longer than previously thought when entrained in freely circulating room air volumes.
COVID-19 fundamentally changed the game in terms of where precautions against infection are needed. Previous novel viruses, including the 2003 SARS-CoV-1 epidemic, were only transmitted by symptomatic individuals.
COVID-19 gained an evolutionary advantage; it can be spread by presymptomatic individuals at rates as high as 75% of that for symptomatically infected individuals. This results in exponential spread of the virus, as individuals infected with the original COVID-19 strain spread the infection to three others on average. The newer delta variant of the coronavirus has proved more infective, spreading on average to five others, which allowed it to overtake the original strain.
As a result of high transmission rates by asymptomatic and presymptomatic individuals, risk mitigation measures are necessary for the entire population, not only around those displaying symptoms.

UVGI’s role in stopping COVID-19 spread
With the emergence of viral mutations such as the delta variant early in 2021, what was urgently needed (in the absence of an effective and widely available vaccine) was a means to deactivate the virus and render it noninfectious. Ideally, such a technology would position engineering mitigation as close as possible to the source of spread — typically those infected individuals actively shedding virus. As it so happened, we have employed just such a technology since the late 19th century: the deliberate and directed dosing of room air volumes with short wave ultraviolet irradiant energy.
In 1933, pioneering American scientist and sanitary engineer William F. Wells demonstrated the ability of this approach, known as ultraviolet germicidal irradiance, to efficiently inactivate infectious airborne microorganisms and prove the concept of infection via the airborne route. In 1937, Wells would subsequently demonstrate the use of UVGI upper-room air volume disinfection protocols to prevent the epidemic spread of measles in suburban Philadelphia day schools where infection outside the school was unlikely.

His research established those infectious pathogens spread via droplets or aerosols were vulnerable to ultraviolet light at wavelengths at or near 253.7 nanometers. Because the maximum absorption wavelength of DNA or RNA genomic material is 260 nm, the organic molecules which make up the DNA and RNA base pairs can strongly absorb short-wave UV light. After irradiation, groups of base pairs form stable dimers that alter the structure of the pathogen’s genomic material sufficiently to interfere with viral or bacterial replication, rendering those cells noninfectious.
UV light’s germicidal effect is highest in the spectral range between 100 to 280 nm, referred to as the UV-C band. The dosage of UV-C irradiant energy needed to degrade the molecular integrity of the viral genome and render the pathogen as noninfectious is measured as “fluence.” The susceptibility of various microbial pathogens to UV fluence can and does vary widely, which is a key concept to consider when designing UVGI systems. Malayeri, et al. have published a comprehensive collection of UV sensitivity and fluence dosage recommendations that covers a wide range of viral, bacterial, protozoan and algal pathogen strains.

The data advises UVGI system designers on the effective fluence for a given log reduction to achieve complete deactivation of the disease-causing pathogen. An equally important consideration is targeting the appropriate log disinfection rate, where UVGI systems are designed to mitigate the growth of plaque-forming units when considering viruses and colony forming units when considering bacteria.
To specify which UVGI system will disinfect upper-room air volumes, engineers typically use a planar two-dimensional analytical approach. This system relies on simplified workflows, which calculate the average UV-C wattage directed into the upper-room air volume at sufficient intensity and for sufficient time to disinfect infectious pathogens that may be present. The CDC/NIOSH publication “Environmental Controls for Tuberculosis” recommends that engineers provide a uniform UVGI field in the upper unoccupied room volume with a UV fluence rate in the range of 30 to 50 microwatts per square centimeter (µW/cm2).
Some ambiguities of the CDC/NIOSH guidance must be addressed. Assumptions of homogeneous, well-mixed air volumes and uniform UV distribution break down when considering actual rooms, especially those large spaces where crowds may be exposed to an infected individual.
The method lacks a calculation procedure for UV-C fluence delivery to the 3D air volume of the room. It also does not account for pathogen susceptibility or the number and movement of infected individuals within the space. Lastly, the method fails to predict the anticipated reduction in risk of infection because of employing UVGI, making it difficult to assess the value of these systems.
Digitally simulating COVID-19 spread
In 2020, engineers and building scientists from the global engineering and consulting firm Arup conducted fundamental research to digitally simulate COVID-19 disinfection utilizing coupled digital ray tracing and computational fluid dynamics. They simulated UV-C irradiance in room air volumes and the resulting pathogen concentrations to accurately calculate the infection risk to individuals using the Wells-Riley Equation.

This research sought to understand whether CFD could provide an accurate representation of pathogen concentrations. Normally, the movement of room air volumes (and the infectious pathogens entrained within) is often too complex to be accurately described by the Wells-Riley model, which assumes the disease-causing bioaerosols are uniformly distributed throughout the room. Because the model examined represents a small space with reasonably uniform concentrations, Arup researchers were able to compare it with mathematical models that describe idealized pathogen concentrations. Survival rate depends on the ventilation rate, UV intensity and the pathogen’s own susceptibility to UV.
The simulation results follow the expected relationship between UVGI intensity, air change rate and pathogen concentration. The results show UVGI to be more effective when air change rates are low because the virus is not being removed by other means and viral particles stay in the irradiant field long enough to ensure their deactivation. Several combinations of UV intensity and ventilation rate, measured in air changes per hour, were modeled.
Steady-state assessments like those above are valid for continuously occupied spaces, but do not account for realistic movement of people. Beggs and Sleigh introduced time in their idealized analytical model, which the team at Arup compared to transient CFD simulations. In addition to the factors previously considered, the new formula contains a term for viral production rate. This model allows people to examine risks that are not constant, such as when the transitions that occur after infected occupants enter and leave a room and demonstrate the amount of time required for various combinations of ventilation and UVGI to remove the viral load from a room.

The final consideration for designers of engineering mitigations to limit the airborne spread of infectious disease is to answer the question: Will the design work as intended? To understand the combined effect of germicidal UV and ventilation rate on each occupant’s infection risk of being infected, Arup engineers modified the Wells-Riley equation to describe the probability of infection based on these factors.
This modification considers exposure time, with longer periods of exposure leading to more infections and uses an equivalent air change rate that incorporates UV levels and pathogen susceptibility. Arup’s quantitative method for describing UVGI efficacy allows us to model the number of new infections we might expect both with and without UVGI and therefore quantify the benefit of UVGI in terms of occupant health.
Upper-room air volume UVGI is an effective tool to limit the spread of airborne infectious pathogens such as Influenza A and COVID-19. Arup’s building scientists, lighting and mechanical engineers developed workflows that allow for the quantification of UVGI efficacy and infection risk in a variety of architectural space types, including spaces that are not well-mixed.

These workflows include radiometric dosing calculations, which can be performed in real time using standard software tools such as Grasshopper or Radiance for larger and more complex spatial geometries. Results of radiometric modeling can be applied to CFD models to determine pathogen concentrations and risk of infection.
Comparing CFD calculations to analytical methods allowed for the prediction of pathogen concentrations within 5% and infection risk within 3%. Modeling demonstrated that the use of upper-room air volume UVGI reduced infection risk by orders of magnitude. In an era of pandemics, lighting, electrical and mechanical engineers will continue to face fundamental challenges to design effective applications for UVGI technology.
The methodologies and evaluation techniques pioneered by Arup’s building scientists and software engineers can provide a high degree of confidence for architects, designers, building owners and facility managers in selecting UVGI technology to protect the health and safety of the public and keep economic enterprises open and viable.




